ATMP WG
Advanced Therapy Medicinal Products
Advanced Therapy Medicinal Products (ATMPs) represent a paradigm shift in the field of healthcare and society, holding the promise of revolutionizing outcomes, care and societal well-being. They also changing the way how concepts of health and disease are seen. ATMPs encompassing gene therapies, cell therapies and tissue-engineering products and describing therapeutical approaches which are standing in contrast to the current healthcare landscape. They offer a personalized approach, addressing the individual genetic attributes and specific needs of patients, thus diverging from conventional therapies that often target broad patient populations. ATMPs target the underlying causes of diseases, potentially leading to more enduring and precise treatments, as opposed to merely alleviating symptoms. This potential faces a range of complex technical, regulatory, and societal implications, warranting a broad examination of their effects on health, healthcare systems and society.
The complexity surrounding Advanced Therapy Medicinal Products (ATMPs) necessitates a collaborative and multidisciplinary approach combined with a proactive way of handling technologies related health needs. International cooperation plays a pivotal role in enhancing the readiness of individuals, healthcare systems and society in terms of ATMPs.
The i-HTS ATMP working group aims to raise awareness, knowledge dissemination, and data accessibility based within the area of ATMPs. The group serves as a platform of shared understanding of needs, requirements and expectations between individuals and organizations with diverse skills and backgrounds and facilitates the exchange of information as well as the development of new methodologies and standards especially within the non-industrial landscape of ATMPs.
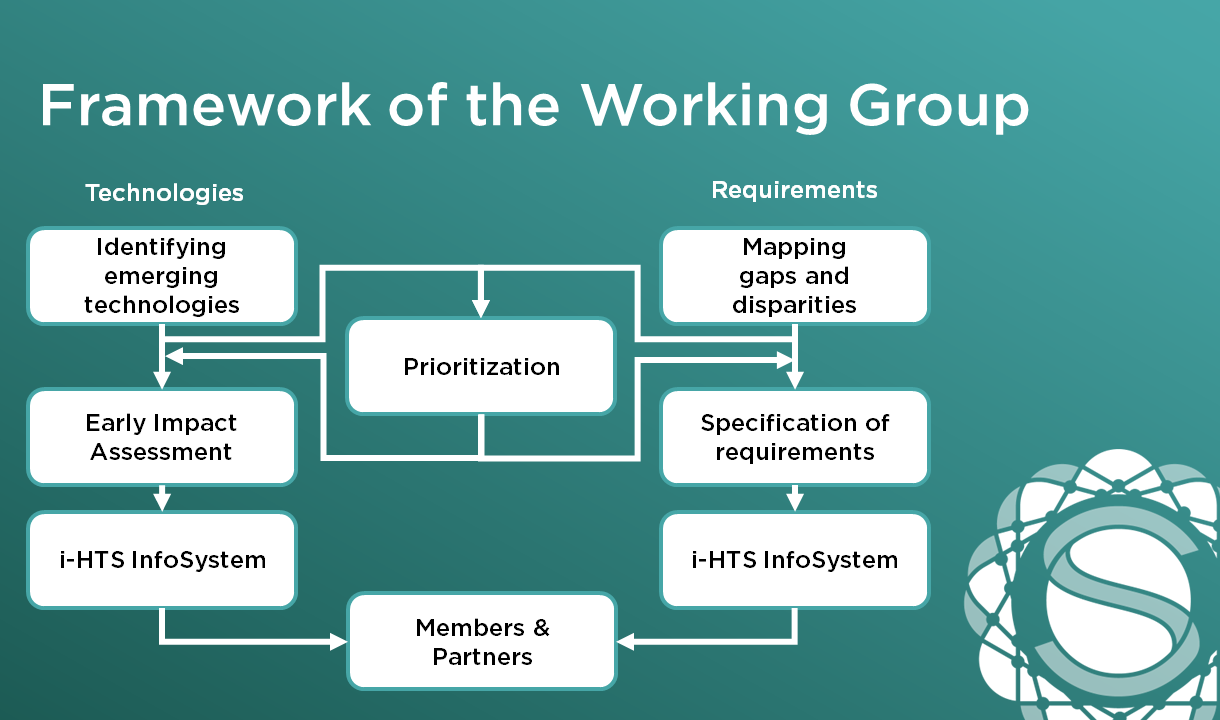
The framework established by the i-HTS ATMP working group serves as a cornerstone for promoting international cooperation, knowledge dissemination and identification of relevant action fields in the area of ATMP for science and policy. This includes:
Raise awareness of ATMPs in the health sector and society.
Identify existing gaps and inequalities in access to ATMPs.
Methods for describing and evaluating ATMPs (based on horizon scanning and health technology assessment) and new requirements (e. g. new infrastructure, training)

